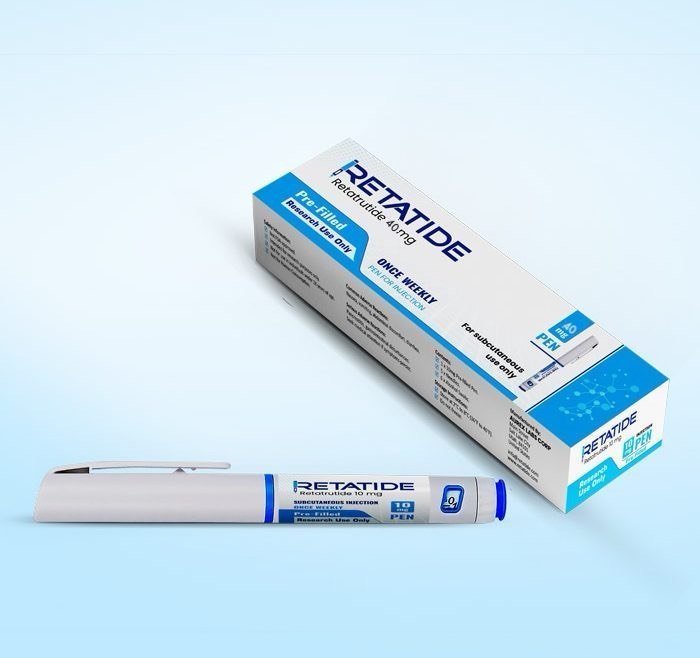
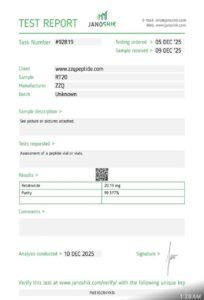
Description
Retatrutide 20MG
Retatrutide (LY3437943) is an investigational triple agonist medication developed by Eli Lilly and Company. It targets three key hormones involved in metabolism and appetite regulation:
- GLP-1 (glucagon-like peptide-1)
- GIP (glucose-dependent insulinotropic polypeptide)
- Glucagon
Unlike dual agonists like tirzepatide (Mounjaro/Zepbound), retatrutide’s triple action has shown superior weight loss in clinical trials—up to 24.2% body weight reduction (about 58 lbs on average) after 48 weeks at the highest dose. It’s primarily studied for:
- Obesity (BMI ≥30 or ≥27 with comorbidities)
- Type 2 diabetes
- Potential benefits in liver fat reduction (MASLD/NAFLD), cardiovascular health, and sleep apnea.
Status: Not FDA-approved yet (as of October 2024). Phase 3 trials (TRIUMPH program) are ongoing, with results expected in 2025. Approval could come in 2026-2027 if successful.
20 mg Dose: What We Know
- Highest studied dose: 20 mg is the maximum from Phase 2 trials (published in NEJM, 2023).
- Administered once weekly via subcutaneous injection (similar to Ozempic/Wegovy).
- Titration: Starts low (e.g., 2-4 mg) and ramps up over 12-20 weeks to minimize side effects.
- Efficacy at 20 mg (Phase 2 data, n=338 adults with obesity):
| Time Point | Weight Loss (%) | HbA1c Reduction (diabetics) |
|---|---|---|
| 24 weeks | 17.5% | -2.02% |
| 48 weeks | 24.2% | -2.02% |
- 58% of participants lost ≥20% body weight.
- Better than semaglutide (15-20%) or tirzepatide (21%) in head-to-head comparisons.
Mechanism Breakdown:
GLP-1: Suppresses appetite, slows gastric emptying
GIP: Enhances insulin secretion, improves fat metabolism
Glucagon: Boosts energy expenditure, preserves muscle mass
= Synergistic fat loss + metabolic boostSide Effects & Safety
Common (dose-dependent, mostly GI):
- Nausea (50-60% at 20 mg), vomiting, diarrhea, constipation.
- Heart rate increase (+11 bpm average).
- Milder than predecessors due to glucagon’s muscle-sparing effects (less lean mass loss: ~25% of total vs. 40% with GLP-1 alone).
Rare/Serious:
- Pancreatitis, gallbladder issues, thyroid tumors (black-box warning like other GLP-1s).
- No increased hypoglycemia risk.
Discontinuation: ~16% at 20 mg (mostly GI intolerance).
| Dose | Nausea Rate | Dropout Rate |
|---|---|---|
| 4 mg | 35% | 7% |
| 12 mg | 52% | 13% |
| 20 mg | 59% | 16% |
Availability & Warnings
- Not commercially available: Only in clinical trials. “20 mg vials” sold online are likely counterfeit/repurposed research chems—avoid them.
- Risks: Incorrect dosing, contamination, unknown purity → hospitalization or death reported with fake semaglutide/tirzepatide.
- Cost estimate (post-approval): $1,000-1,500/month (like Zepbound).
- Compounding pharmacies: Some offer “retatrutide” now, but unverified and illegal without approval.
How to Get It Legally
- Join a clinical trial (clinicaltrials.gov: search “retatrutide”).
- Wait for approval—monitor Lilly’s updates.
- Alternatives now: Tirzepatide (21% loss), semaglutide (15-20%).
Consult a doctor before any weight loss drug. Not for cosmetic use; screen for contraindications (e.g., medullary thyroid cancer history).





Reviews
There are no reviews yet.